Research is one of the core initiatives of the University of Wisconsin – Madison Department of Radiology. Providing the necessary infrastructure and resources for investigators to be successful is key in supporting this initiative, and so the Department Research and Development Committee has awarded several pilot grants to projects. Take a look below at some of the exciting work that’s been funded by the Department of Radiology!
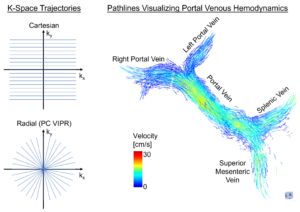
4D Flow MRI – Paving the way for clinical application: Comparing the two most popular acquisition methods
Principal Investigators: Andrew P Huang, (medical student) and Thekla H. Oechtering, MD
Radiology Mentor: Scott Reeder, MD PhD
Overview: 4D Flow MRI is a powerful technique for non-invasive visualization and measurement of blood flow in all three spatial directions, resolved in time. The characterization of hemodynamics in the liver is a promising application potentially allowing risk stratification in patients with end-stage liver disease. This is a research focus at UW in the labs of Dr. Scott Reeder, Dr. Alejandro Roldán-Alzate, Dr. Oliver Wieben and Dr. Kevin Johnson. The R&D funded “CaRad” project (Cartesian vs Radial 4D Flow MRI) is comparing the two most popular methods to capture 4D flow MRI data. The two methods are “Cartesian” encoding that collects data in k-space line-by-line to fill up an area shaped like a cube and “radial” encoding that collects data via spokes radiating from a central point to fill an area shaped like a sphere. The radial method was developed here at UW and is called “PC-VIPR” (phase contrast vastly undersampled isotropic voxel radial projection imaging). It is significantly faster than traditional Cartesian methods, acquiring more data with higher temporal and spatial resolution. Vendor-provided 4D flow sequences typically use Cartesian sampling and are a robust method in a non-research setting.
By comparing the two methods, we will evaluate whether the results from both sequences are reproducible – an important prerequisite for broad clinical application. Therefore, we will acquire 4D Flow MRI in healthy volunteers and patients using both methods on the same day. We will compare 4D flow measurements, as well as test-retest repeatability, and image quality.
Artificial Intelligence (AI) Tool for Conversion of MRI to PET
Principal Investigators: Vivek Prabhakaran, MD, PhD and Vikas Singh, PhD
Overview: An estimated 6.2 million Americans ages 65 and older are living with Alzheimer’s dementia (AD) in 2021, which is estimated to increase to 14 million by 2050 (Alzheimer’s Association, 2021). There is a growing urgency to diagnose AD, as well as precursor stages, so patients can be appropriately managed clinically. MRI, Amyloid PET, and 18F-fluorodeoxyglucose ([18F]FDG)-PET, are all utilized for clinically diagnosing dementia. The use of artificial intelligence (AI) in the study of AD has the potential to develop a reliable way to generate a synthetic brain PET scan from a MR image. The study will use a large sample (N=3000) of retrospective T1-, T2-, ASL perfusion, DTI weighted MR and PET images (Amyloid, FDG), collected in the clinical and research settings from healthy subjects and those with AD and Mild Cognitive Impairment. MRI images (training n=2700) will be used to train and validate an AI-based Convolutional Neural Network to produce synthetic PET images and compared with ground truth PET images. The trained model will be tested for diagnostic performance using a test set (testing n=300). Successful ability to convert MRI to PET would be a major paradigm shift in the field of medicine, which could lead to significant cost savings and allow for more rapid, efficient, and noninvasive imaging workup of dementia.
Appearance of the Post-Operative Acetabular Labrum and Articular Cartilage on Magnetic Resonance Imaging to Include T2* Cartilage Mapping: A Prospective Study with Imaging at 1-week, 6-months & 1-year
Principal Investigator: Donna Blankenbaker, MD, FACR
Project Lead: Zachary Stewart, MD
Overview: This project, a collaboration with the Department of Radiology and the Department of Orthopedic Surgery, aims to clarify what the normal post-operative acetabular labrum looks like on MRI in attempt to improve diagnostic accuracy when re-imaging these patients. Currently, there is a general consensus, both anecdotal and supported by the literature, that the imaging characteristics of the re-torn labrum and the normal post-operative labrum have overlapping imaging characteristics that one would not expect to see in the normal non-instrumented labrum. Prior studies have not evaluated the normal early post-operative appearance of the labrum, and it is our hope that by doing so we can better characterize the expected post-surgical changes to differentiate those features from that of new/recurrent tears. Additionally, acetabular cartilage delamination frequently occurs in conjunction with acetabular labral tears but is notoriously challenging to image and can also be difficult for the surgeon to see arthroscopically. Utilizing a relatively new imaging technique, T2* cartilage mapping, we aim to better define the role of this imaging sequence in this patient population and characterize the T2* appearance of the post-operative acetabular cartilage.